Periodate /pəˈraɪ.ədeɪt/ is an anion composed of iodine and oxygen. It is one of a number of oxyanions of iodine and is the highest in the series, with iodine existing in oxidation state +7. Unlike other perhalogenates, such as perchlorate, it can exist in two forms: metaperiodate IO−
4 and orthoperiodate IO5−
6. In this regard it is comparable to the tellurate ion from the adjacent group. It can combine with a number of counter ions to form periodates, which may also be regarded as the salts of periodic acid.
Periodates were discovered by Heinrich Gustav Magnus and C. F. Ammermüller; who first synthesised periodic acid in 1833.[1]
Synthesis
Classically, periodate was most commonly produced in the form of sodium hydrogen periodate (Na3H2IO6).[2] This is commercially available, but can also be produced by the oxidation of iodates with chlorine and sodium hydroxide.[3] Or, similarly, from iodides by oxidation with bromine and sodium hydroxide:
Modern industrial scale production involves the electrochemical oxidation of iodates, on a lead dioxide (PbO2) anode, with the following standard electrode potential:
Metaperiodates are typically prepared by the dehydration of sodium hydrogen periodate with nitric acid,[2] or by dehydrating orthoperiodic acid by heating it to 100 °C under vacuum.
They can also be generated directly from iodates by treatment with other strong oxidizing agents such as hypochlorites:
Forms and interconversion
Periodate can exist in a variety of forms in aqueous media, with pH being a controlling factor. Orthoperiodate has a number of acid dissociation constants.[5][6]
- pKa = 3.29
- pKa = 8.31
- pKa = 11.60
The ortho- and metaperiodate forms also exist in equilibrium.
- K = 29
For this reason orthoperiodate is sometimes referred to as the dihydrate of metaperiodate,[7] written IO−4·2H2O; however, this description is not strictly accurate as X-ray crystallography of H5IO6 shows 5 equivalent I−OH groups.[8]
At extremes of pH additional species can form. Under basic conditions a dehydration reaction can take place to form the diperiodate (sometimes referred to as mesoperiodate).
- K = 820
Under strongly acid conditions periodic acid can be protonated to give the orthoperiodonium cation.[9]
- pKa = −0.8
Structure and bonding
In both the ortho- and metaperiodate the iodine is hypervalent, as it forms more bonds than would classically be allowed. This has been explained in terms of dative bonds, confirming the absence of double bonding in these molecules.[10]
Exact structures vary depending on counter ions, however on average orthoperiodates adopt a slightly deformed octahedral geometry with X-ray diffraction showing I–O bond lengths of 1.89 Å.[11][8] Metaperiodates adopt a distorted tetrahedral geometry with an average I–O distance of 1.78 Å.[12][13]
Reactions
Cleavage reactions
Periodates can cleave carbon–carbon bonds on a variety of 1,2-difunctionalised alkanes.[14][15] The most common example of this is diol cleavage, which was also the first to be discovered (Malaprade reaction).[16] In addition to diols, periodates can cleave 1,2-hydroxy ketones, 1,2-diketones, α-keto acids, α-hydroxy acids, amino acids,[17] 1,2-amino alcohols,[18] and 1,2-diamines,[19] to give aldehydes, ketones, and carboxylic acids. In the presence of strong acid catalyst, like H2SO4 or HNO3 epoxides[20][21] are also converted into aldehyde or ketones or dicarbonyl compounds.

Alkenes can also be oxidised and cleaved in the Lemieux–Johnson oxidation. This uses a catalytic loading of osmium tetroxide which is regenerated in situ by the periodate. The overall process is equivalent to that of ozonolysis.
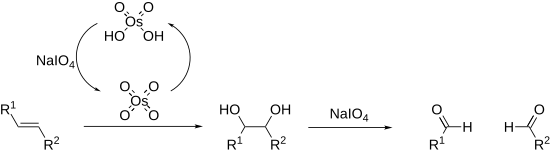
Cleavage reactions proceed via a cyclic intermediate called a periodate ester. The formation of this may be affected by pH and temperature[22] but is most strongly affected by the geometry of the substrate, with cis-diols reacting significantly faster than trans-diols.[23] The reactions are exothermic and are typically performed at 0 °C. As periodate salts are only readily soluble in water reactions are generally performed in aqueous media. Where solubility is an issue periodic acid may be used, as this is soluble in alcohols; phase transfer catalysts are also effective in biphasic reaction mixtures. In extreme cases the periodate may be exchanged for lead tetraacetate which reacts in a similar manner and is soluble in organic solvents (Criegee oxidation).

Periodate cleavage is often utilized in molecular biochemistry for the purposes of modifying saccharide rings, as many five- and six-membered sugars have vicinal diols. Historically it was also used to determine the structure of monosaccharides.[24][25]
Periodate cleavage may be performed on an industrial scale to form dialdehyde starch which has uses in paper production.[26]
Oxidation reactions
Periodates are powerful oxidising agents. They can oxidise catechol to 1,2-benzoquinone and hydroquinone to 1,4-benzoquinone.[27] Sulfides can be effectively oxidised to sulfoxides.[28] Periodates are sufficiently powerful to generate other strong inorganic oxidisers such as permanganate,[29] osmium tetroxide[30] and ruthenium tetroxide.
Niche uses
Periodates are highly selective etchants for certain ruthenium-based oxides.[31]
Several staining agents use in microscopy are based around periodate (e.g. periodic acid–Schiff stain and Jones' stain).
Periodates have also been used as oxidising agents for use in pyrotechnics.[32] In 2013 the US Army announced that it would replace the environmentally harmful chemicals barium nitrate and potassium perchlorate with sodium metaperiodate for use in their tracer ammunition.[33]
https://en.wikipedia.org/wiki/Periodate
Production
Sulfur dioxide is primarily produced for sulfuric acid manufacture (see contact process). In the United States in 1979, 23.6 million metric tons (26 million U.S. short tons) of sulfur dioxide were used in this way, compared with 150,000 metric tons (165,347 U.S. short tons) used for other purposes. Most sulfur dioxide is produced by the combustion of elemental sulfur. Some sulfur dioxide is also produced by roasting pyrite and other sulfide ores in air.[17]
https://en.wikipedia.org/wiki/Sulfur_dioxide
Cosmetics
The safety of cosmetic products is constantly in question as the components are always changing or being discovered as a possibly harmful substance. The sulfite components of cosmetic ingredients, such as sodium bisulfite, underwent clinical trials to find out their safety in cosmetic formulations. Sodium bisulfite functions as a reducing agent and furthermore as a hair-waiving/straightening agent.[8] As of 1998, sodium bisulfite was used in 58 different cosmetic products including hair conditioners, moisturizers, and hair dyes.[9]
In a cosmetic context, the reducing ability of sodium bisulfite is used to prevent discoloration, bleach food starches, and delay spoilage of the product. Since the sulfite molecule was used in so many compounds in the 1990s, the EPA, FDA, and American Conference of Governmental Industrial Hygienists established a working place threshold limit value for sulfur dioxide of 2ppm averaged over 8 hours, and a 3-hour level of 5ppm. Even with this threshold established, the FDA recognized sodium bisulfite as a "generally recognized as safe" compound.[3]
A final examination on the carcinogenicity, genotoxicity, oral toxicity, and cellular toxicity on consumed sodium bisulfite was conducted using living subjects such as mice and rats. The International Agency for Research on Cancer concluded that there was inadequate evidence that sodium bisulfite was carcinogenic.[3] Under specific conditions such as acidity and concentration level, sodium bisulfite was able to cause negative alterations to the genome such as catalyzing transamination, and induce sister-chromatid exchanges suggesting possible genotoxicity.[10] In a study using Osbourne-Mendel strain rats, it was concluded that oral toxicity was not significant if the consumed concentration was less than 0.1% (615ppm as SO2).[11] A study done by Servalli, Lear, and Cottree in 1984 found that sodium bisulfite did not produce membrane fusion in hepatic and murine glial cells and human fibroblasts so there is no oral toxicity. These clinical studies concluded that sodium bisulfite was safe to use in cosmetic formulations.[3]
https://en.wikipedia.org/wiki/Sodium_bisulfite
Sodium croscarmellose is an internally cross-linked sodium carboxymethylcellulose for use as a superdisintegrant in pharmaceutical formulations.[1]
E468 is the E number of crosslinked sodium carboxymethyl cellulose, used in food as an emulsifier.
https://en.wikipedia.org/wiki/Sodium_croscarmellose
Background
The cross-linking reduces water solubility while still allowing the material to swell (like a sponge) and absorb many times its weight in water. As a result, it provides superior drug dissolution and disintegration characteristics, thus improving formulas′ subsequent bioavailability by bringing the active ingredients into better contact with bodily fluids.
Sodium croscarmellose also resolves formulators′ concerns over long-term functional stability, reduced effectiveness at high tablet hardness levels, and similar problems associated with other products developed to enhance drug dissolution. It is a very commonly used[2] pharmaceutical additive approved by the U.S. Food and Drug Administration. Its purpose in most tablets – including dietary supplements – is to assist the tablet in disintegrating in the gastrointestinal tract promptly. If a tablet disintegrating agent is not included, the tablet could disintegrate too slowly, in the wrong part of the intestine or not at all, thereby reducing the efficacy and bioavailability of the active ingredients.
Croscarmellose is made by first soaking crude cellulose in sodium hydroxide, and then reacting the cellulose with sodium monochloroacetate to form sodium carboxymethylcellulose. Excess sodium monochloroacetate slowly hydrolyzes to glycolic acid and the glycolic acid catalyzes the cross-linkage to form sodium croscarmellose.[1] Chemically, it is the sodium salt of a cross-linked, partly O-(carboxymethylated) cellulose.
Sodium croscarmellose was first used as a stabilizer in horse supplements.[citation needed]
https://en.wikipedia.org/wiki/Sodium_croscarmellose
https://en.wikipedia.org/wiki/Category:Excipients
 Liquid paraffin in a bottle | |
| Clinical data | |
|---|---|
| Routes of administration | Topical, oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem SID | |
| ChemSpider |
|
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.029.438 |
| Chemical and physical data | |
| Formula | C nH 2n+2 |
Liquid paraffin, also known as paraffinum liquidum, paraffin oil, liquid paraffin oil or Russian mineral oil, is a very highly refined mineral oil used in cosmetics and medicine. Cosmetic or medicinal liquid paraffin should not be confused with the paraffin (i.e. kerosene) used as a fuel. The generic sense of paraffin meaning alkane led to regional differences for the meanings of both paraffin and paraffin oil. It is a transparent, colorless, nearly odorless, and oily liquid that is composed of saturated hydrocarbons derived from petroleum.[1]
The term paraffinum perliquidum is sometimes used to denote light liquid paraffin, while the term paraffinum subliquidum is sometimes used to denote a thicker mineral oil.
https://en.wikipedia.org/wiki/Liquid_paraffin_(drug)

A vaccine dose contains many ingredients, very little of which is the active ingredient, the immunogen. A single dose may have merely nanograms of virus particles, or micrograms of bacterial polysaccharides. A vaccine injection, oral drops or nasal spray is mostly water. Other ingredients are added to boost the immune response, to ensure safety or help with storage, and a tiny amount of material is left-over from the manufacturing process. Very rarely, these materials can cause an allergic reaction in people who are very sensitive to them.
Volume

The volume of a vaccine dose is influenced by the route of administration. While some vaccines are given orally or nasally, most require an injection. Vaccines are not injected intravenously into the bloodstream. Most injections deposit a small dose into a muscle, but some are given superficially just under the skin surface or deeper beneath the skin.[1]
Fluenz Tetra, a live flu vaccine for children, is administered nasally with 0.1ml of liquid sprayed into each nostril. The live typhoid vaccine, Vivotif,[2] and a live adenovirus vaccine, licensed only for military use, both come as hard gastro-resistant tablets.[3] The Sabin oral live polio vaccine is taken as two 0.05ml drops of a bitter salty liquid that was historically added to sugar cubes when given to young children.[4] Rotarix, a live rotavirus vaccine, has about 1.5ml of liquid containing 1g of sugar to make it taste better.[5] The Dukoral cholera vaccine comes as a 3ml suspension along with 5.6g of effervescent granules, which are mixed and added to around 150ml water to make a sweet raspberry flavoured drink.[6]
At the other end of the volume scale, the smallpox vaccine is a minuscule 0.0025ml droplet that is picked up when a bifurcated needle is dipped into a vial containing around 100 doses. This needle is pricked 15 times into a small area of skin, just firmly enough to produce a drop of blood.[7] A little larger is the BCG tuberculosis vaccine, which is 0.05ml for babies and children under 12, and 0.1ml for others. This tiny dose is inserted a couple of millimetres under the skin, producing a small blanched blister.[8] Many vaccines for intramuscular injection have 0.5ml liquid, though a few have 1ml.[a]
Some vaccines come with the active ingredients already suspended in solution and the syringe pre-filled (e.g., Bexsero meningococcal Group B vaccine[10]). Others are supplied as a vial of freeze-dried powder, which is reconstituted prior to administration using a dilutant from a separate vial or pre-filled syringe (e.g., MMR vaccine[13]). Infanrix hexa, the 6-in-1 vaccine that protects against six diseases, uses a combination approach: the Hib vaccine in the powder and DTPa-HBV-IPV in suspension.[9] Alternatively two separate vaccine solutions are mixed just before administration (ViATIM hepatitis A and typhoid vaccine).[19]
Immunogens
Many vaccines developed in the 20th century contain whole bacteria or viruses, which are either inactivated (killed), attenuated (weakened) or a strain chosen to be harmless in humans. Since these are so small, even a tiny amount of them contains a huge number of individuals.[21]
With bacterial vaccines, we can enumerate this with an approximate number of bacteria cells. The live typhoid vaccine contains two billion viable cells of Salmonella enterica subsp. enterica serovar Typhi, which have been attenuated and cannot cause disease.[2] The cholera vaccine has over thirty billion of each of four strains of Vibrio cholerae, which are inactivated by heat or formalin.[6] The BCG vaccine, infant dose, contains between 100,000 and 400,000 colony-forming unit of live attenuated Mycobacterium bovis.[8]
One way to count viruses is to observe their impact on host cells in tissue cultures. The two tablets of adenovirus vaccine, one with adenovirus type 4 and the other with type 7, each contain 32,000 tissue-culture infective doses (104.5 TCID50).[3] The current live polio vaccine contains two serotypes of poliovirus: over 1 billion tissue-culture infective doses (106 TCID50) of type 1 and over 630,000 (105.8 TCID50) of type 3.[4] The smallpox vaccine contains between 250,000 and 1,250,000 plaque forming units of live vaccina virus per dose.[7] The MMR vaccine contains 1,000 TCID50 measles, 12,500 TCID50 mumps and 1,000 TCID50 rubella live attenuated viruses.[13]
Many modern vaccines are made of only the parts of the pathogen necessary to invoke an immune response (a subunit vaccine) – for example just the surface proteins of the virus, or only the polysaccharide coating of a bacterium. Some vaccines invoke an immune response against the toxin produced by bacteria, rather than the bacteria itself. These toxoid vaccines are used against tetanus, diphtheria and pertussis (whooping cough). If the bacteria polysaccharide coating produces only a weak immune response on its own, it may be combined with (carried on) a protein that does provoke a strong response, which in turn improves the response to the weaker component. Such conjugate vaccines, may make use of a toxoid as the carrier protein.[21] For all these, the quantity of immunogen is given by weight and sometimes expressed as international units (IU). The HVP vaccine contains 120 micrograms of the L1 capsid proteins from four types of human papillomavirus.[15] The pneumococcal conjugate vaccine contains 32 micrograms of pneumococcal polysaccharide conjugated with CRM197 (a diphtheria toxin).[11]
Another variant is the RNA vaccine, which contains mRNA embedded in lipid (fat) nanoparticles. The mRNA instructs body's own cell machinery to produce the proteins that stimulate the immune response.[21] Comirnaty, the Pfizer-BioNTech COVID-19 vaccine contains thirty micrograms of BNT162b2 RNA.[22]
Excipients
Excipients are substances present in the vaccine that are not the principal immunological agents. These may be present to enhance the vaccine's potency, ensure safety, aid with storage or are left over from the manufacturing process.[23]
Adjuvants
Live vaccines produce a strong immune response that lasts a long time, but they are not suitable for people with weakened immune systems. Other kinds of vaccine, where the pathogen has been inactivated or that contain only part of the pathogen, often alone produce a weaker response and require booster doses. In these vaccines, a substance called an adjuvant is added to make the immune response stronger and longer lasting.[21][24]
The most commonly used adjuvants are aluminium salts such as aluminium hydroxide, aluminium phosphate or potassium aluminium sulphate (also simply called alum).[24][25] These aluminium salts can be responsible for soreness and redness at the vaccination site but do not cause any long-term harm to human health. The amount of aluminium in these vaccines ranges from 0.125 milligrams in the pneumococcal conjugate vaccine to 0.82 milligrams in the 6-in-1 vaccine. The Meningococcal Group B vaccine contains 0.5 milligrams and in the UK Immunisation Schedule is given at the same time as the 6-in-1 vaccine at eight and sixteen weeks, giving a combined dose of 1.32 milligrams of aluminium. Aluminium salts are commonly and naturally consumed in small quantities, and the quantity in this combined vaccine dose is lower than the weekly safe intake level.[25] Vaccines containing aluminium adjuvants cannot be frozen or allowed to freeze accidentally in a refrigerator, as this causes the particles to coagulate and damages the antigen.[26]
Another adjuvant used in some flu vaccines is an oil-in-water emulsion. The oil, squalene, is found in all plant and animal cells, and is commercially extracted and purified from shark liver. The flu vaccine for older adults, Fluad, uses an adjuvant branded MF59, which has squalene (9.75 milligrams), citric acid (0.04 milligrams) and three emulsifiers: polysorbate 80, sorbitan trioleate, sodium citrate (1.175, 1.175 and 0.66 milligrams respectively).[27] The H1N1 swine-flu vaccine, Pandemrix, used the adjuvant branded AS03, which has squalene (10.69 milligrams), DL-α-tocopherol (11.86 milligrams) and polysorbate 80 (4.86 milligrams)[28]
Preservatives
Preservatives prevent the growth of bacteria and fungi, and are more commonly used in vaccines produced as multi-dose vials. They must also be non-toxic in the dose used and not adversely affect the immunogenicity of the vaccine.[29] Thiomersal is the best known and most controversial preservative. It was phased out of UK vaccines between 2003 and 2005 and is not used in any routine vaccines in the UK. As a precaution, the US and Europe have also removed thiomersal from vaccines, despite there being no evidence of harm.[25] The US-licensed vaccines in the routine paediatric schedule generally have no thiomersal at all; a few have only a trace amount as a residual from manufacturing (less than one microgram). This is also the case for influenza vaccines in the US that come in single-dose vials or prefilled syringes. Some influenza vaccines are also available as a multi-dose vial, and in that form contain thiomersal (24.5 micrograms of mercury).[30]
Phenol 0.25% v/v is used in Pneumovax 23, a pneumococcal polysaccharide vaccine, and in the smallpox vaccine. However, phenol reduces the potency of diphtheria and tetanus toxoid-containing vaccines. Similarly, thiomersal weakens the immunogenicity of the inactivated poliovirus vaccine, so the IPOL vaccine contains 2–3 microlitres of 2-phenoxyethanol instead.[31][32]
Stabilisers
Stabilisers protect the vaccine from the effects of temperature and ensure it does not degrade in storage. For vaccines that are freeze-dried, they provide a necessary bulk. Without them, the vaccine powder would be invisibly tiny (ranging from nanograms to a few tens of micrograms) and stick to the vial glass. Stabilisers used for vaccines include sugars (sucrose, lactose), sorbitol, amino acids (glycine, monosodium glutamate) and proteins (hydrolysed gelatin). There have very rarely (one in two million vaccinations) been cases of allergic reaction to the proteins in gelatin.[25][33] The source of gelatin, pork, is of religious concern to Jewish and Muslim communities, though some leaders have ruled this is not a cause to reject vaccines that are injected or inhaled rather than ingested. There are alternatives for some vaccines that contain gelatine.[25][34]
Acidity regulators such as phosphate salts keep the pH within a required range during manufacture and in the final product. Other salts help ensure the vaccine is isotonic with body fluids.[35]
Manufacturing residuals
There are materials that serve no function in the final vaccine but are left over from the manufacturing process. Bacteria and viruses may be inactivated using formaldehyde. The quantity remaining in diphtheria or tetanus toxoid vaccines licensed in the US is required to be less than 0.1 milligrams (0.02%). Although formaldehyde has potentially toxic and carcinogenic properties in large doses, it is present in the blood (due to natural biochemical processes) at much higher concentrations than permitted in vaccines. Alternatives used in some vaccines include glutaraldehyde and β-propiolactone. Antibiotics may be used to prevent bacteria growing during vaccine manufacture and traces of these may remain. Antibiotics that some people are allergic to (such as cephalosporins, penicillins and sulphonamides) are not used. Those that are used include kanamycin, gentamicin, neomycin, polymyxin B, and streptomycin.[25][36]
A small amounts of protein may remain from the material used to grow viruses, to which some people may be hypersensitive. Some influenza and yellow fever vaccines are grown in chicken eggs, and measles or mumps vaccines may be grown in chick embryo cell culture. Engerix-B, a recombinant DNA vaccine for hepatitis B is produced in yeast and may contain up to five percent yeast protein.[36] Cervarix, an HPV vaccine, is grown in a cell line from the cabbage looper moth.[37] The amount of insect protein remaining is less than forty nanograms.[36]
Some components of the vaccine vial or syringe may contain latex rubber. This is a problem for those with a severe allergic reaction to latex, but not for those who get contact dermatitis after wearing latex gloves.[25][38]
Notes
https://en.wikipedia.org/wiki/Vaccine_ingredients
| |
| Names | |
|---|---|
| IUPAC name
Magnesium octadecanoate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.320 |
| E number | E572 (acidity regulators, ...) |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
|---|---|
| Mg(C 18H 35O 2) 2 | |
| Molar mass | 591.27 g/mol |
| Appearance | light white powder |
| Odor | slight |
| Density | 1.026 g/cm3 |
| Melting point | 88.5 °C (191.3 °F; 361.6 K) |
| 0.003 g/100 mL (15 °C) 0.004 g/100 mL (25 °C) 0.008 g/100 mL (50 °C) | |
| Solubility | negligible in ether and alcohol slightly soluble in benzene |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 250 °C (482 °F; 523 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
> 1000 mg/kg (oral, rat) |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
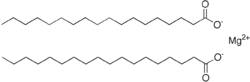
Magnesium stearate is the chemical compound with the formula Mg(C
18H
35O
2)
2. It is a soap, consisting of salt containing two equivalents of stearate (the anion of stearic acid) and one magnesium cation (Mg2+).
Magnesium stearate is a white, water-insoluble powder. Its
applications exploit its softness, insolubility in many solvents, and
low toxicity. It is used as a release agent and as a component or lubricant in the production of pharmaceuticals and cosmetics.[1]
https://en.wikipedia.org/wiki/Magnesium_stearate

A plasticizer (UK: plasticiser) is a substance that is added to a material to make it softer and more flexible, to increase its plasticity, to decrease its viscosity, and/or to decrease friction during its handling in manufacture.
Plasticizers are commonly added to polymers such as plastics and rubber, either to facilitate the handling of the raw material during fabrication, or to meet the demands of the end product's application. For example, plasticizers are commonly added to polyvinyl chloride (PVC), which otherwise is hard and brittle, to make it soft and pliable; which makes it suitable for products such as shower curtains, vinyl flooring, clothing, bags, flexible plastic tubing, and electric wire insulation/coating.
Plasticizers are also often added to concrete formulations to make them more workable and fluid for pouring, thus allowing the water contents to be reduced. Similarly, they are often added to clays, stucco, solid rocket fuel, and other pastes prior to molding and forming. For these applications, plasticizers largely overlap with dispersants.
https://en.wikipedia.org/wiki/Plasticizer













No comments:
Post a Comment